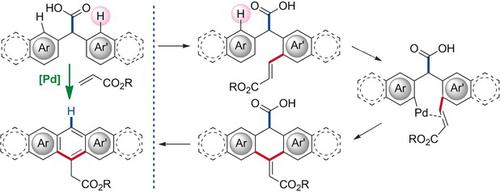
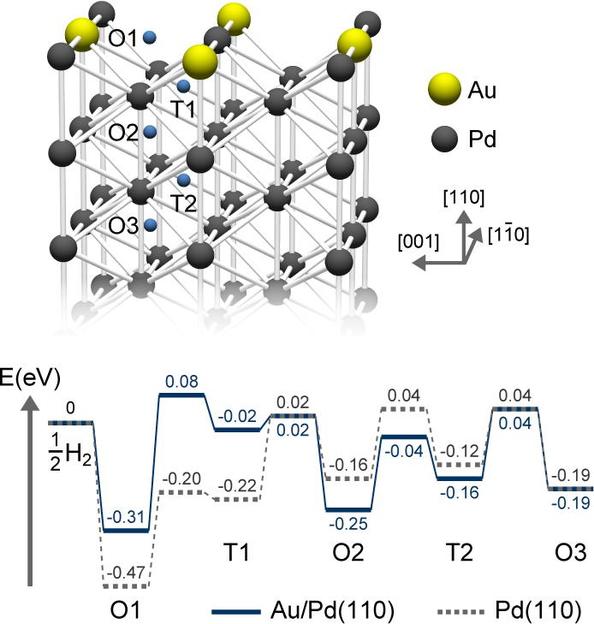
Directed C−H Activation and Tandem Cross‐Coupling Reactions Using Palladium Nanocatalysts with Controlled Oxidation - Angew. Chem. Int. Ed. - X-MOL
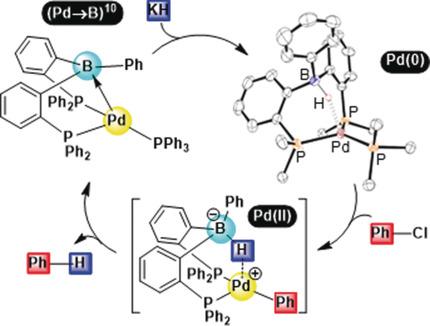
Palladium-Borane Cooperation: Evidence for an Anionic Pathway and Its Application to Catalytic Hydro-/Deutero-dechlorination. - Angew. Chem. Int. Ed. - X-MOL
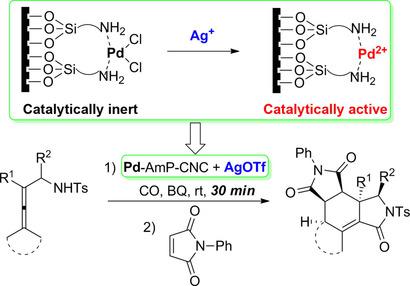
Silver-Triggered Activity of a Heterogeneous Palladium Catalyst in Oxidative Carbonylation Reactions. - Angew. Chem. Int. Ed. - X-MOL
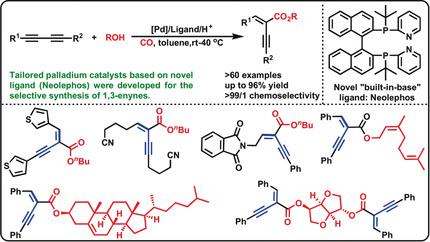
Tailored Palladium Catalysts for Selective Synthesis of Conjugated Enynes by Monocarbonylation of 1,3-Diynes.,Angewandte Chemie International Edition - X-MOL

Organometallic Chemistry: Palladium-Catalyzed Arylation of Carbasugars Enables the Discovery of Potent and Selective SGLT2 Inhibitors - Ng - 2016 - Angewandte Chemie International Edition - Wiley Online Library
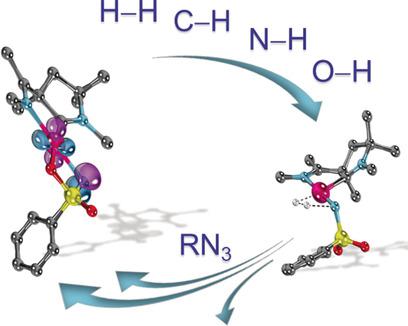
An Isolable Terminal Imido Complex of Palladium and Catalytic Implications,Angewandte Chemie International Edition - X-MOL

Frontispiz: An Electron‐Poor C64 Nanographene by Palladium‐Catalyzed Cascade C−C Bond Formation: One‐Pot Synthesis and Single‐Crystal Structure Analysis - Seifert - 2016 - Angewandte Chemie - Wiley Online Library

Unveiling the Local Structure of Palladium Loaded into Imine‐Linked Layered Covalent Organic Frameworks for Cross‐Coupling Catalysis - Romero‐Muñiz - 2020 - Angewandte Chemie - Wiley Online Library
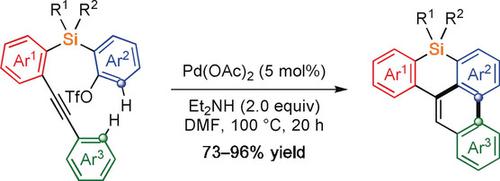
Palladium-Catalyzed Synthesis of Benzophenanthrosilines by C-H/C-H Coupling through 1,4-Palladium Migration/Alkene Stereoisomerization.,Angewandte Chemie International Edition - X-MOL
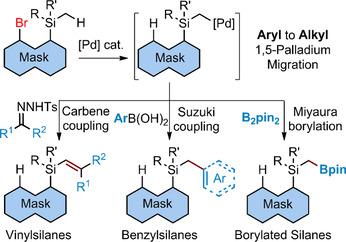
Divergent Synthesis of Vinyl-, Benzyl-, and Borylsilanes: Aryl to Alkyl 1,5- Palladium Migration/Coupling Sequences. - Angew. Chem. Int. Ed. - X-MOL
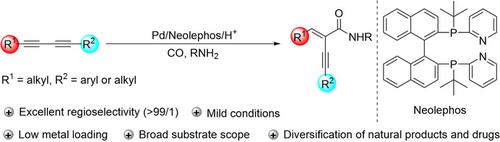
A General and Highly Selective Palladium-Catalyzed Hydroamidation of 1,3-Diynes.,Angewandte Chemie International Edition - X-MOL

Palladium(II)‐Initiated Catellani‐Type Reactions - Cheng - 2019 - Angewandte Chemie - Wiley Online Library