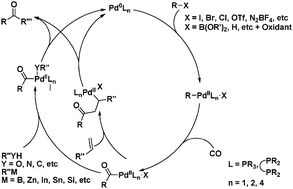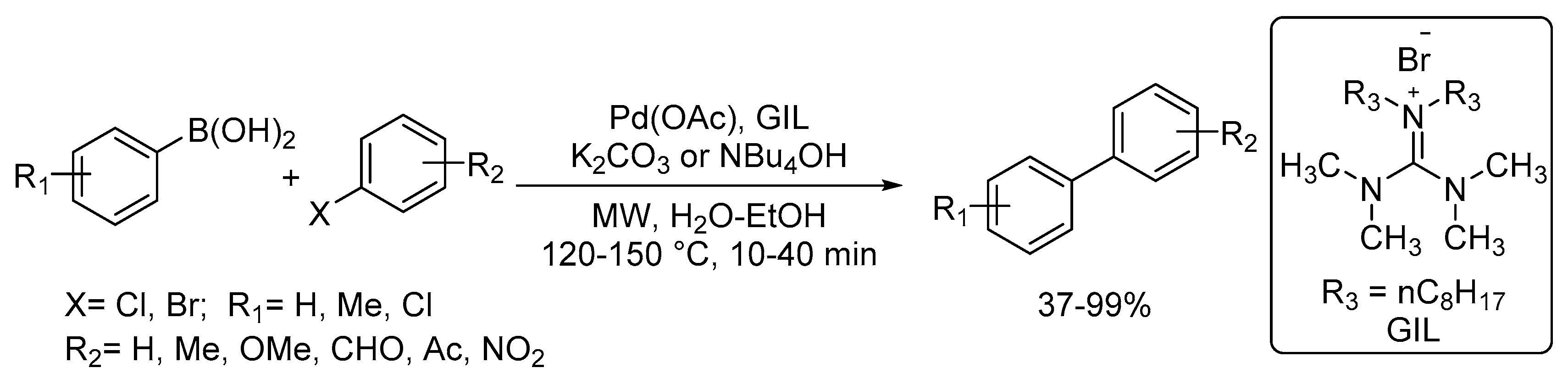
Catalysts | Free Full-Text | Microwave-Assisted Palladium-Catalyzed Cross-Coupling Reactions: Generation of Carbon–Carbon Bond | HTML

Recent Progress in the Use of Pd-Catalyzed C-C Cross-Coupling Reactions in the Synthesis of Pharmaceutical Compounds

Recent Progress in the Use of Pd-Catalyzed C-C Cross-Coupling Reactions in the Synthesis of Pharmaceutical Compounds

Recent Progress in the Use of Pd-Catalyzed C-C Cross-Coupling Reactions in the Synthesis of Pharmaceutical Compounds
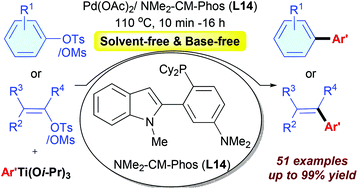
Palladium-catalyzed cross-coupling of (hetero)aryl or alkenyl sulfonates with aryl titanium as the multi-functional reagent - Organic Chemistry Frontiers (RSC Publishing)
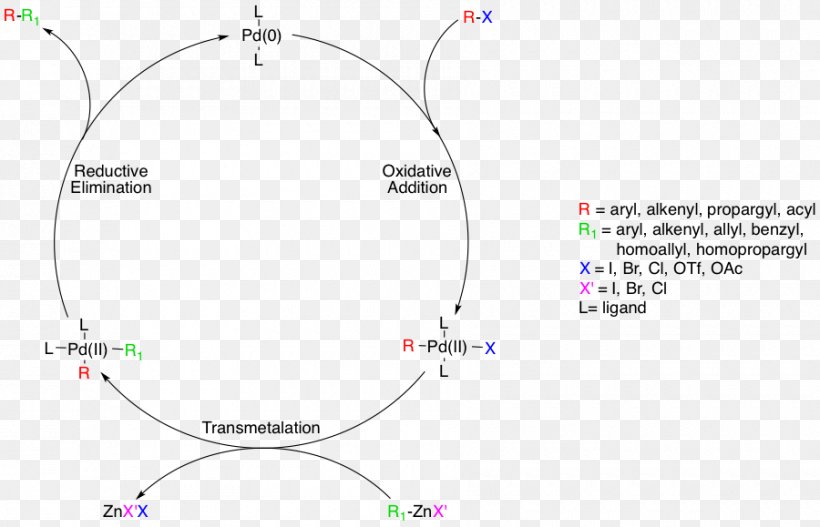
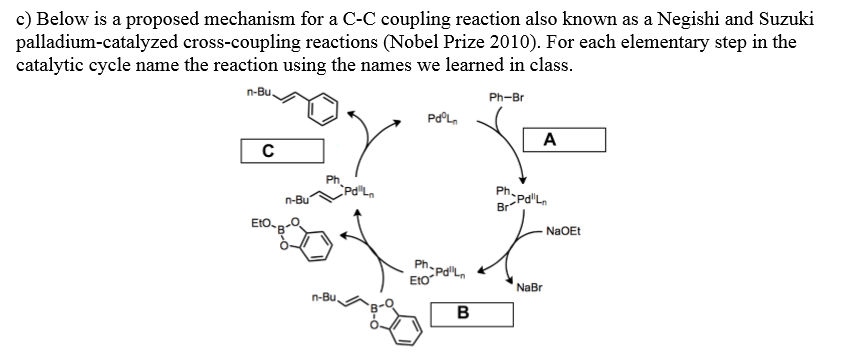
Negishi Coupling Palladium-catalyzed Coupling Reactions Reaction Mechanism Oxidative Addition, PNG, 900x579px, Negishi Coupling, Akira Suzuki,

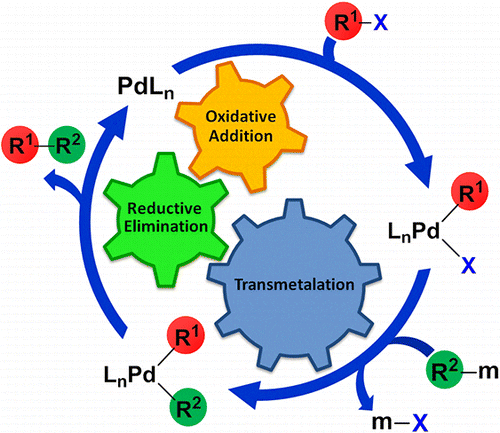
Recent Advances in Pd‐Catalyzed Cross‐Coupling Reaction in Ionic Liquids - Li - 2018 - European Journal of Organic Chemistry - Wiley Online Library

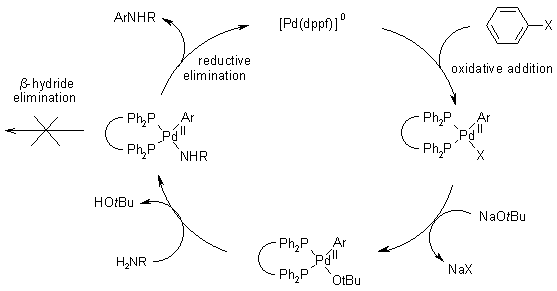
Functionalized nitrogen ligands for palladium catalyzed cross-coupling reactions (part I) - ScienceDirect
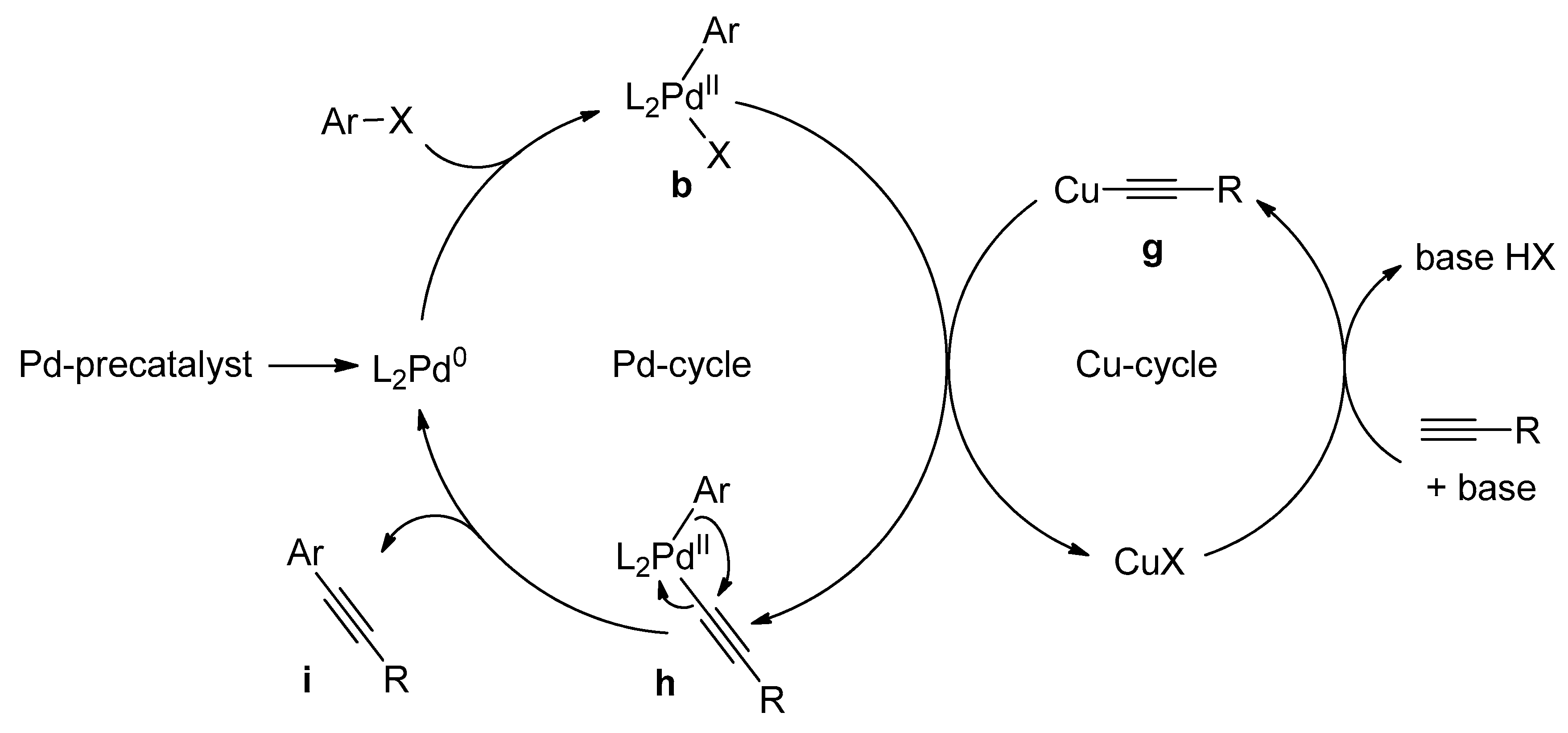
Catalysts | Free Full-Text | Microwave-Assisted Palladium-Catalyzed Cross-Coupling Reactions: Generation of Carbon–Carbon Bond | HTML
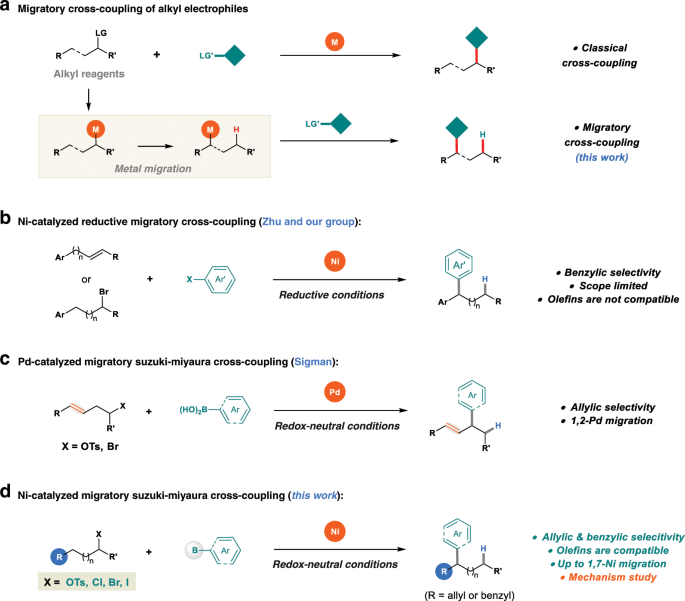
Reaction scope and mechanistic insights of nickel-catalyzed migratory Suzuki–Miyaura cross-coupling | Nature Communications
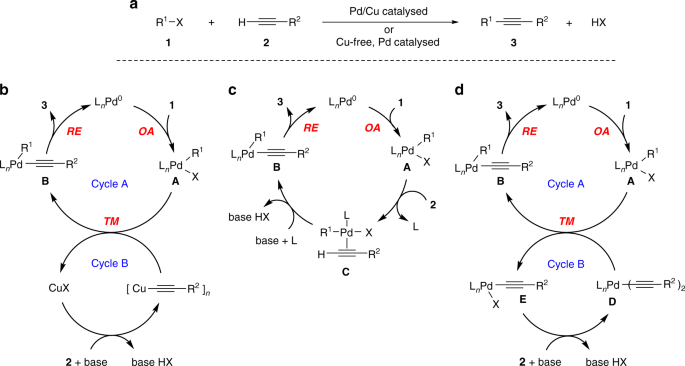
Mechanism of copper-free Sonogashira reaction operates through palladium- palladium transmetallation | Nature Communications
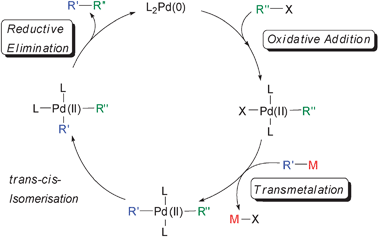
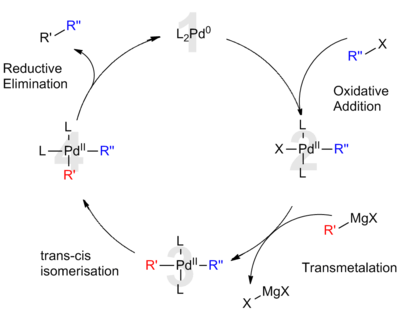
35 years of palladium -catalyzed cross-coupling with Grignard reagents : how far have we come? - Chemical Society Reviews (RSC Publishing) DOI:10.1039/C1CS15137A