The predictive power of aromaticity: quantitative correlation between aromaticity and ionization potentials and HOMO–LUMO gaps in oligomers of benzene, pyrrole, furan, and thiophene - Physical Chemistry Chemical Physics (RSC Publishing)
![PDF] Electrostatic considerations affecting the calculated HOMO-LUMO gap in protein molecules. | Semantic Scholar PDF] Electrostatic considerations affecting the calculated HOMO-LUMO gap in protein molecules. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/60222a314a0dbd061c570b3d64e7dcde6ab79a10/5-Figure1-1.png)
PDF] Electrostatic considerations affecting the calculated HOMO-LUMO gap in protein molecules. | Semantic Scholar

Unraveling the Role of Π - Conjugation in Thiophene Oligomers for Optoelectronic Properties by DFT/TDDFT Approach

SOLVENT EFFECT ON THE STRUCTURAL, ELECTRONIC, SPECTRA PROPERTIES AND FIRST HYPERPOLARIZABILITY OF W(CO)5L, L=(4-PYRIDYLMETHYLENE)MALONONITRILE

Rational design of near‐infrared absorbing organic dyes: Controlling the HOMO–LUMO gap using quantitative molecular orbital theory - Narsaria - 2018 - Journal of Computational Chemistry - Wiley Online Library

Communication: Highest occupied molecular orbital–lowest unoccupied molecular orbital gaps of doped silicon clusters from core level spectroscopy: The Journal of Chemical Physics: Vol 134, No 4

Variation of the HOMO−LUMO gap with increasing conjugation length of... | Download Scientific Diagram
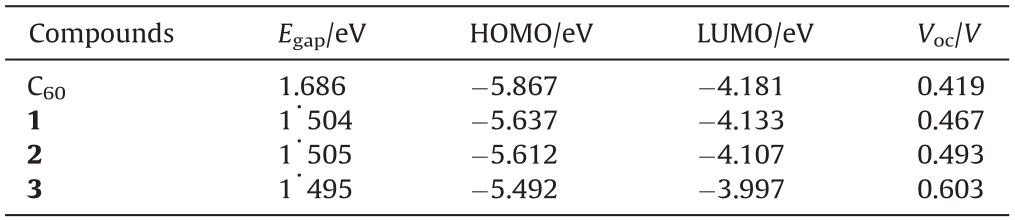
Effect of the position of substitution on the electronic properties of nitrophenyl derivatives of fulleropyrrolidines:Fundamental understanding toward raising LUMO energy of fullerene electron-acceptor
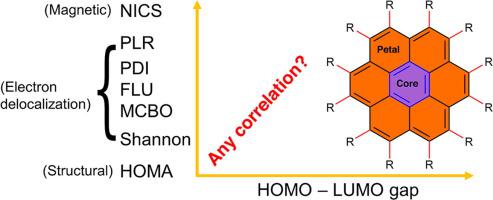
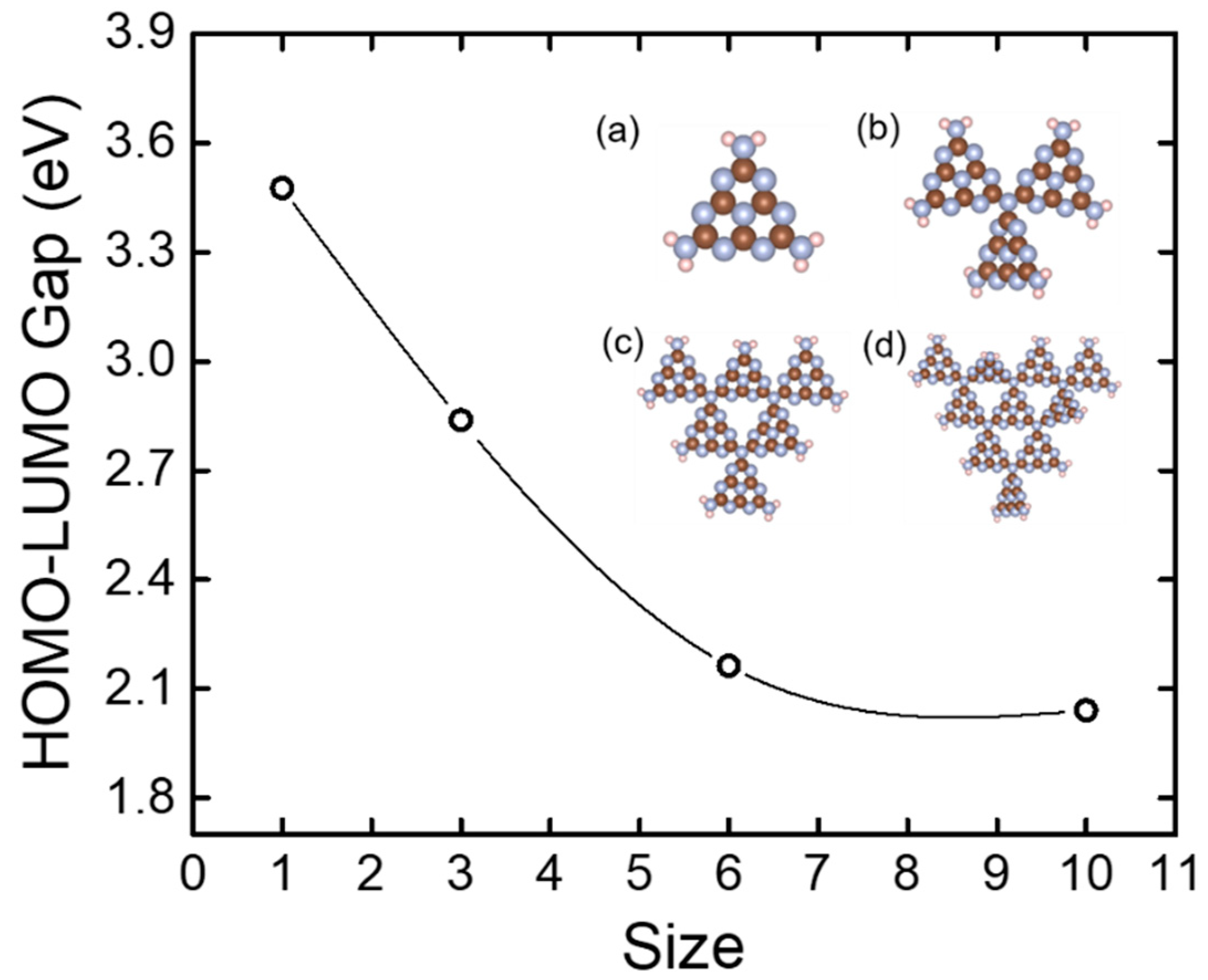
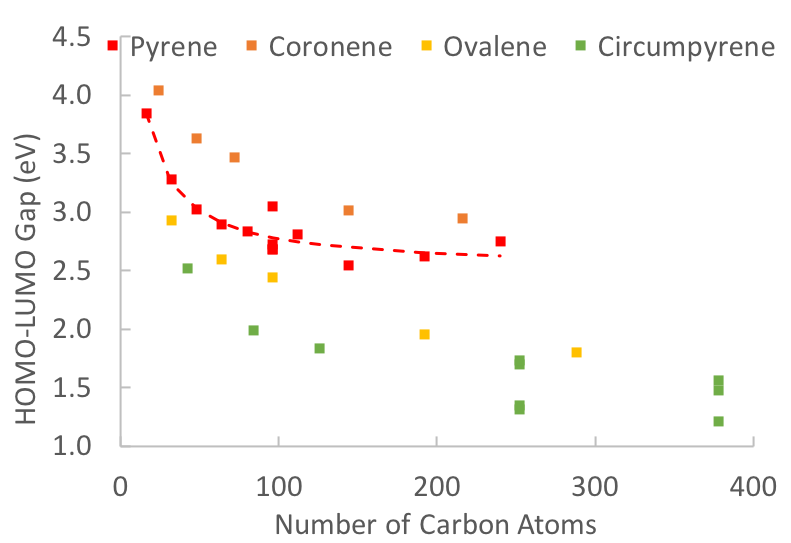
How do magnetic, structural, and electronic criteria of aromaticity relate to HOMO – LUMO gap? An evaluation for graphene quantum dot and its derivatives - Chem. Phys. - X-MOL

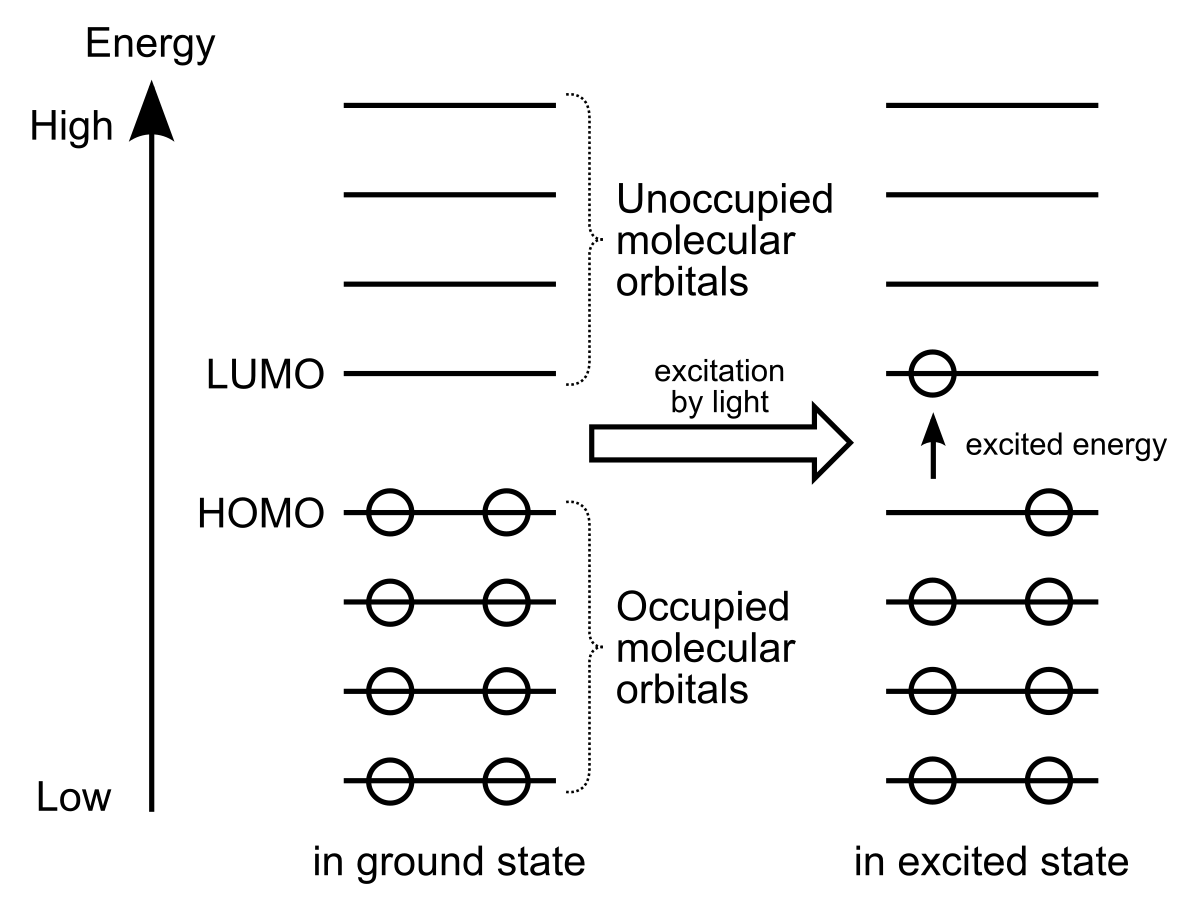
Molecular orbital energy diagram, HOMO-LUMO energy gaps and β tot (×... | Download Scientific Diagram

An unusual mechanism for HOMO–LUMO gap narrowing in a minimal near-IR dye generated by the deprotonation of bis(dicyanomethylene)indan - ScienceDirect