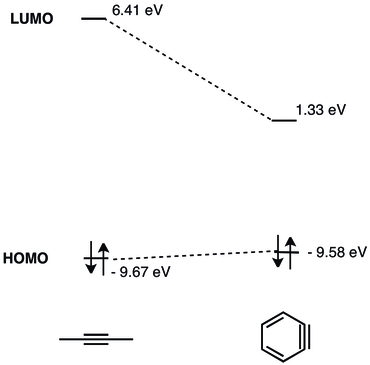
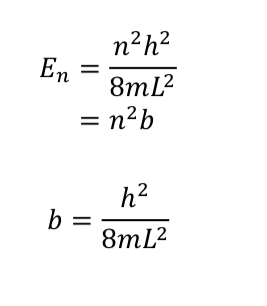HOMO–LUMO Energy-Gap Tuning of π-Conjugated Zwitterions Composed of Electron-Donating Anion and Electron-Accepting Cation | The Journal of Organic Chemistry

Figure 4 | Synthesis, Crystal Structure, DFT Calculations and Hirshfeld Surface Analysis of 3-Chloro-3-methyl-r(2),c(6)-bis(p-methoxyphenyl)piperidin-4-one | SpringerLink

Tuning the HOMO–LUMO Energy Gap of Small Diamondoids Using Inverse Molecular Design | Journal of Chemical Theory and Computation

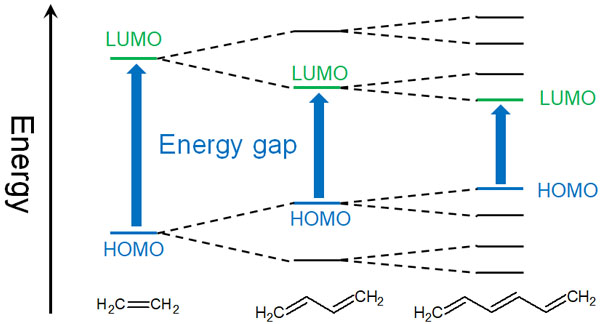
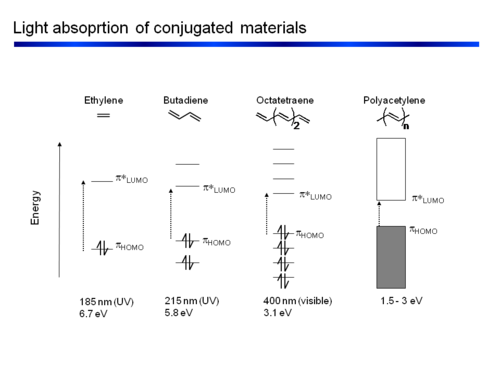
Variation of the HOMO−LUMO gap with increasing conjugation length of... | Download Scientific Diagram
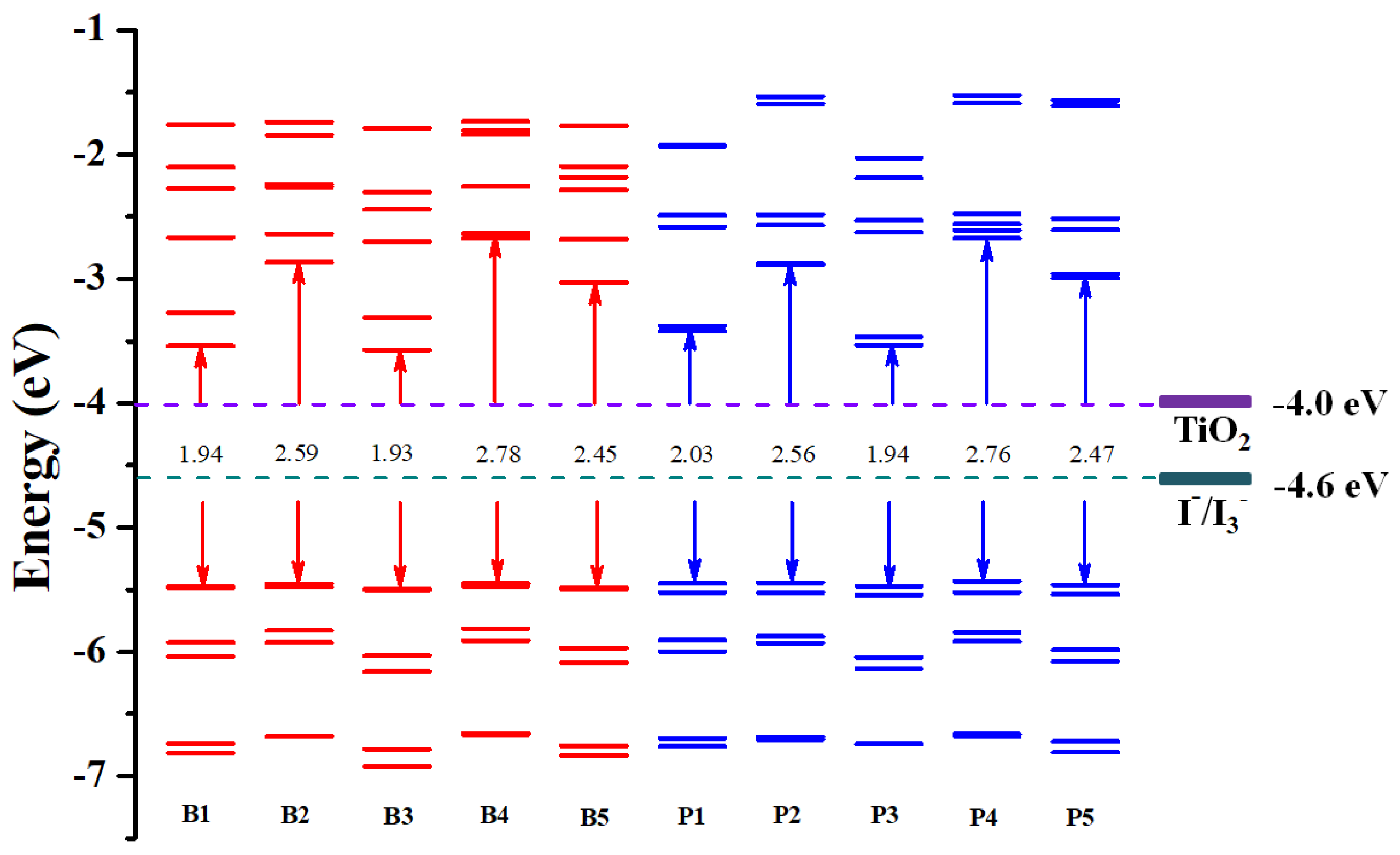
Molecules | Free Full-Text | Theoretical Analysis on Heteroleptic Cu(I)-Based Complexes for Dye-Sensitized Solar Cells: Effect of Anchors on Electronic Structure, Spectrum, Excitation, and Intramolecular and Interfacial Electron Transfer | HTML

Molecular orbital energy diagram, HOMO-LUMO energy gaps and β tot (×... | Download Scientific Diagram

The solvent (water) and metal effects on HOMO-LUMO gaps of guanine base pair: A computational study - ScienceDirect

HOMO–LUMO Energy-Gap Tuning of π-Conjugated Zwitterions Composed of Electron-Donating Anion and Electron-Accepting Cation | The Journal of Organic Chemistry



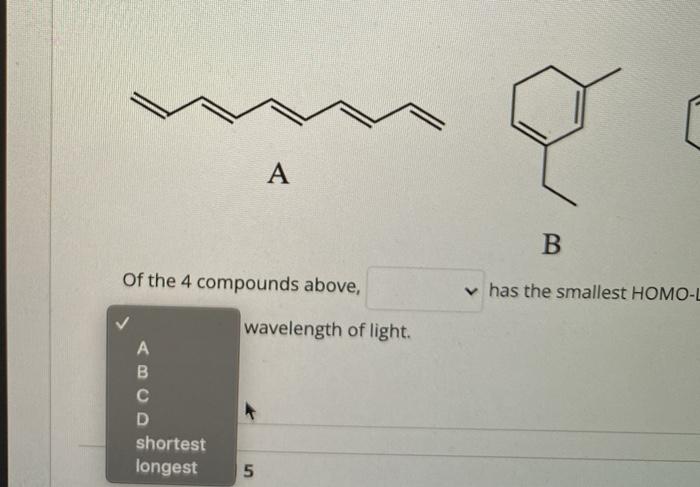


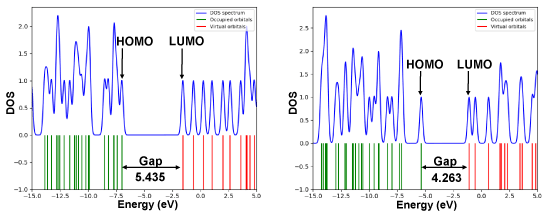
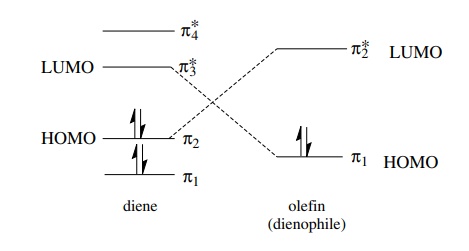

/chapter4/pages9and10/page9_10_files/ethanehl.png)