Торговые центры Праги: Палладиум, Ходов, Котва, Метрополь. Торговые центры в праге Торговый центр в праге

Neutral and cationic (pyrazolylmethyl)pyridine palladium(II) complexes: kinetics and chemoselectivity studies in hydrogenation of alkenes and alkynes | SpringerLink
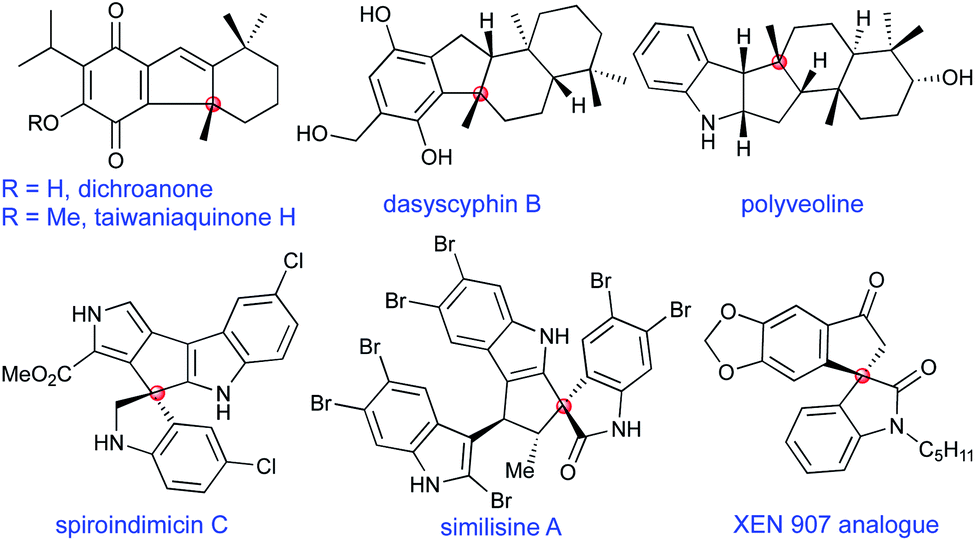
Palladium-catalysed 5- endo-trig allylic (hetero)arylation - Chemical Science (RSC Publishing) DOI:10.1039/D0SC01932A

Bimetallic Pd( III ) complexes in palladium-catalysed carbon–heteroatom bond formation | Nature Chemistry
![Palladium, [1,4,8,11,15,18,22,25‐Octabutoxy‐29H,31H‐phthalocyaninato(2‐)‐κN29,κN30,κN31,κN32]‐, (SP‐4‐1)‐ - Ravetz - - Major Reference Works - Wiley Online Library Palladium, [1,4,8,11,15,18,22,25‐Octabutoxy‐29H,31H‐phthalocyaninato(2‐)‐κN29,κN30,κN31,κN32]‐, (SP‐4‐1)‐ - Ravetz - - Major Reference Works - Wiley Online Library](https://onlinelibrary.wiley.com/cms/asset/71822bce-d4a0-4b5d-a1a4-2b11de664100/047084289x.cover.jpg)
Palladium, [1,4,8,11,15,18,22,25‐Octabutoxy‐29H,31H‐phthalocyaninato(2‐)‐κN29,κN30,κN31,κN32]‐, (SP‐4‐1)‐ - Ravetz - - Major Reference Works - Wiley Online Library

Aerobic Palladium(II)‐Catalyzed 5‐endo‐trig Cyclization: An Entry into the Diastereoselective C‐2 Alkenylation of Indoles with Tri‐ and Tetrasubstituted Double Bonds - Kandukuri - 2012 - Angewandte Chemie International Edition - Wiley Online Library
![Palladium, [1,4,8,11,15,18,22,25‐Octabutoxy‐29H,31H‐phthalocyaninato(2‐)‐κN29,κN30,κN31,κN32]‐, (SP‐4‐1)‐ - Ravetz - - Major Reference Works - Wiley Online Library Palladium, [1,4,8,11,15,18,22,25‐Octabutoxy‐29H,31H‐phthalocyaninato(2‐)‐κN29,κN30,κN31,κN32]‐, (SP‐4‐1)‐ - Ravetz - - Major Reference Works - Wiley Online Library](https://onlinelibrary.wiley.com/cms/asset/1bb8368e-cbb3-49e3-b210-b93819ade6e1/nrn02305es.001.gif)